Evaluating Scope and Trends for the Base Cation Decline in Surface Waters in the Northeastern US
In 1984, the U.S. Environmental Protection Agency assessed the chemistry of eastern U.S. lakes. In the Northeast, 145 of the lakes were re-sampled in 1986 by EPA and again in 2004 by NSRC researchers, who assessed biologically-relevant chemical changes related to recent reductions in acidic deposition. In particular, they evaluated the significance of the widely reported reduction in base cations (acid neutralizing elements such as calcium and magnesium) in surface waters.
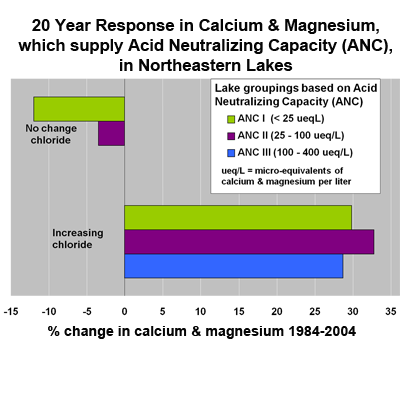
Over 90% of the lakes decreased in sulfur dioxide and aluminum from 1984 to 2004. Changes in concentrations of calcium and magnesium were related to recovery from acidic deposition and affected by lake chloride concentration, a reflection of soil ion exchange reactions related to run-off of road salt (sodium chloride). Approximately half the lakes were unaffected by road salt. Over 70% of the lakes not affected by salt experienced a decrease in calcium and magnesium over 20 years. In contrast, 85% of salt-affected lakes increased in calcium and magnesium.
Reductions in sulfur emissions have been associated with increased acid neutralizing capacity and reduced aluminum concentrations in the most acid sensitive surface waters, both of which are important indicators of improvement in biologically-relevant chemistry in northeastern lakes. However, decreases in base cation concentrations, particularly in low acid neutralizing capacity systems unaffected by road salt, slowed recovery from acidic deposition in some regions. Human-derived sources of salt throughout the northeastern U.S. complicated assessments of changes in lake chemistry by increasing calcium and magnesium in contrast to what appears to be the typical response to declining sulfur deposition.
